A recent study by Sarah Fawaz and colleagues, led by Professor Maria Kaisar at the University of Oxford, used samples and data from the QUOD Biobank to investigate whether inflammation in deceased donors contributes to kidney injury before transplantation and affects graft outcomes afterwards. The study focused on tumour necrosis factor alpha (TNFα) and its receptors, TNFR1 and TNFR2, exploring both their clinical associations and their biological relevance in the donor kidney.
Kidney transplantation remains the best treatment for end-stage kidney disease, but the continued shortage of donor organs means that transplant teams increasingly rely on kidneys from deceased donors, which have more complex risk profiles. While routine donor assessment captures clinical factors such as age and comorbidity, it does not fully reflect the biological stress experienced by organs during donor management. In donation after brain death, this stress can include a pronounced inflammatory response — sometimes described as a cytokine storm — which may contribute to kidney injury even before the organ is retrieved.
To investigate this, the team quantified TNFα, TNFR1, and TNFR2 in 1,018 longitudinal plasma samples collected during donor management from 596 deceased donors (QUOD Biobank) and 34 living donors (Oxford Transplant Biobank). These measurements were linked to recipient outcomes, including graft function at 12 months and long-term graft survival up to 96 months. The study also incorporated matched donor kidney biopsies and mechanistic experiments in cultured human podocytes, allowing the findings in donor plasma to be examined alongside direct evidence of tissue injury.
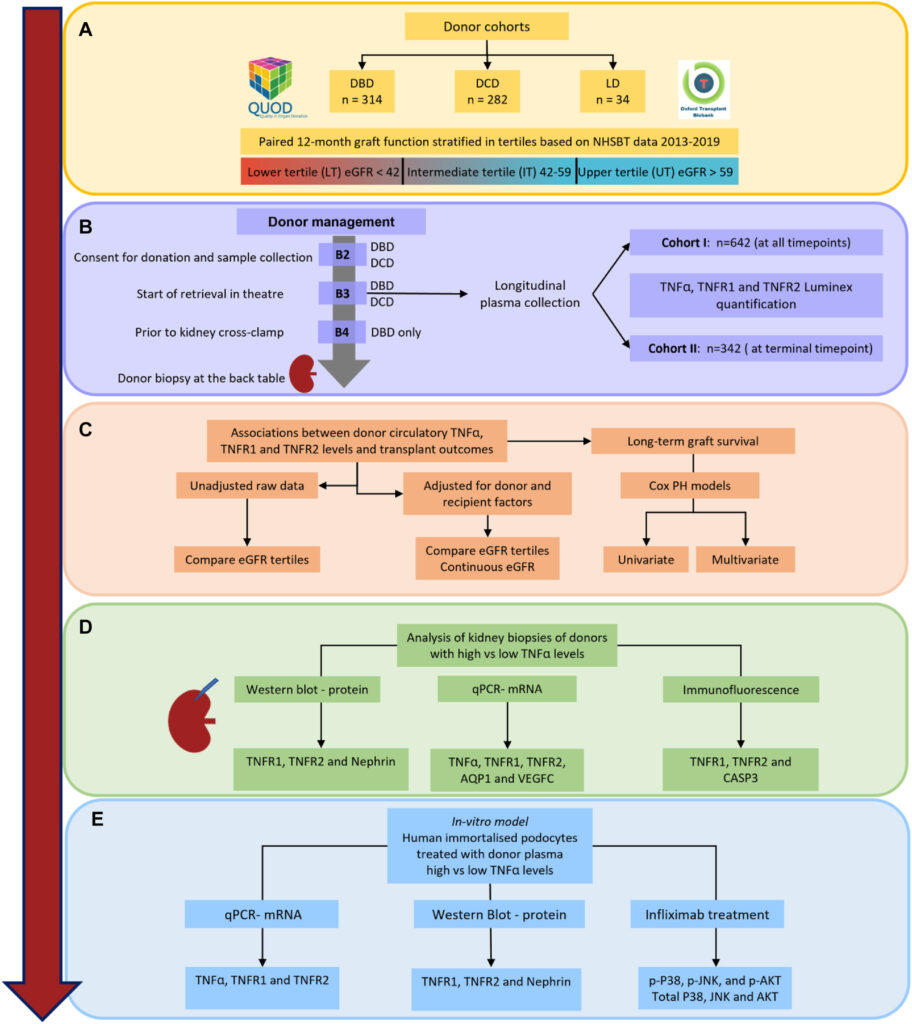
The study found that high donor plasma TNFα levels were strongly associated with poorer kidney graft function at 12 months and up to 60 months, as well as reduced graft survival up to 96 months. Importantly, this signal was seen in donors after brain death, but not in donation after circulatory death, suggesting that the inflammatory biology of brain death may have consequences for kidney quality. These associations were confirmed in an independent validation cohort and remained robust after adjustment for donor and recipient variables.
Analysis of matched kidney biopsies showed that kidneys with high TNFα had increased expression of inflammatory and injury-related markers, including TNFR1, TNFR2, and caspase-3, together with reduced nephrin and aquaporin-1. This suggests damage affecting both the glomeruli and tubules. In vitro experiments strengthened this link further: podocytes exposed to plasma from donors with high TNFα developed an injury-associated profile, including activation of injury-related pathways, such as p38 MAPK and JNK signalling. This response was reduced by infliximab, which blocks TNFα, suggesting that TNFα-driven injury pathways may be not only measurable during donor management, but also potentially modifiable.
Together, these findings highlight donor inflammation as an important determinant of transplant outcome and suggest that inflammatory profiling during donor management could improve donor organ assessment. More broadly, the findings point to a clinically actionable window before transplantation, where targeting pathways, such as TNFα signalling, may help protect donor kidneys and improve long-term graft outcomes.
Summary
Using QUOD Biobank donor plasma and kidney biopsy samples, this study showed that high donor TNFα levels are linked to kidney injury and poorer transplant outcomes, particularly in donation after brain death. By combining clinical associations, biopsy findings, and podocyte experiments, the findings suggests that donor inflammation is both biologically relevant and potentially modifiable before transplantation.
These findings were published in the American Journal of Transplantation Feb 2026: DOI: 10.1016/j.ajt.2026.01.023.
