Sample collection
QUOD samples are collected from donors at a number of timepoints in the organ donation pathway as indicated:
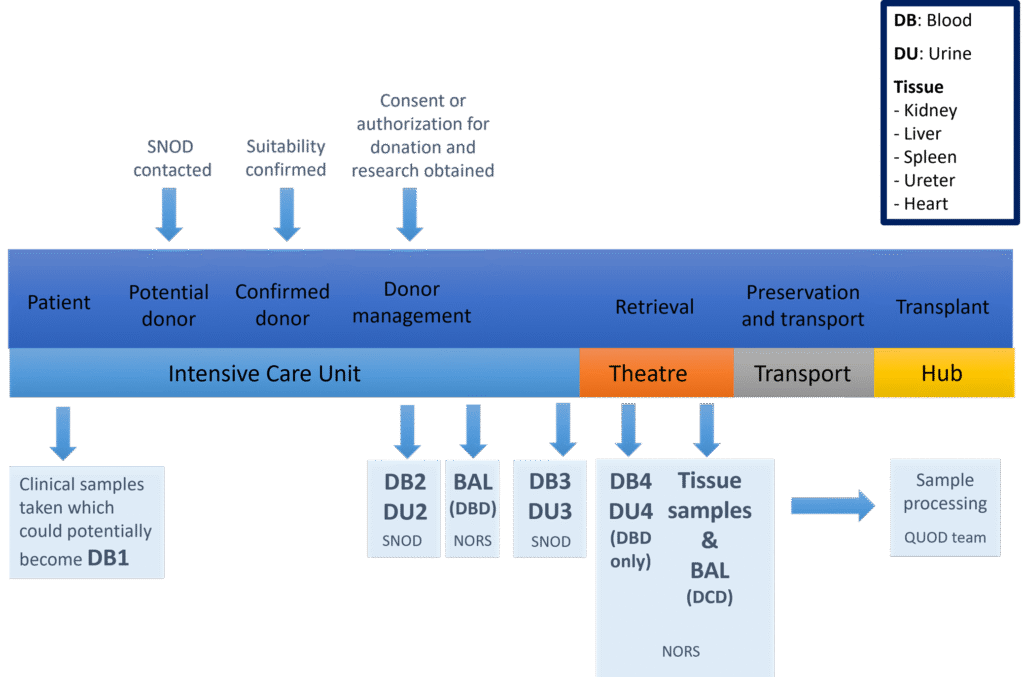
Available samples
The table below summarises the types of samples available.
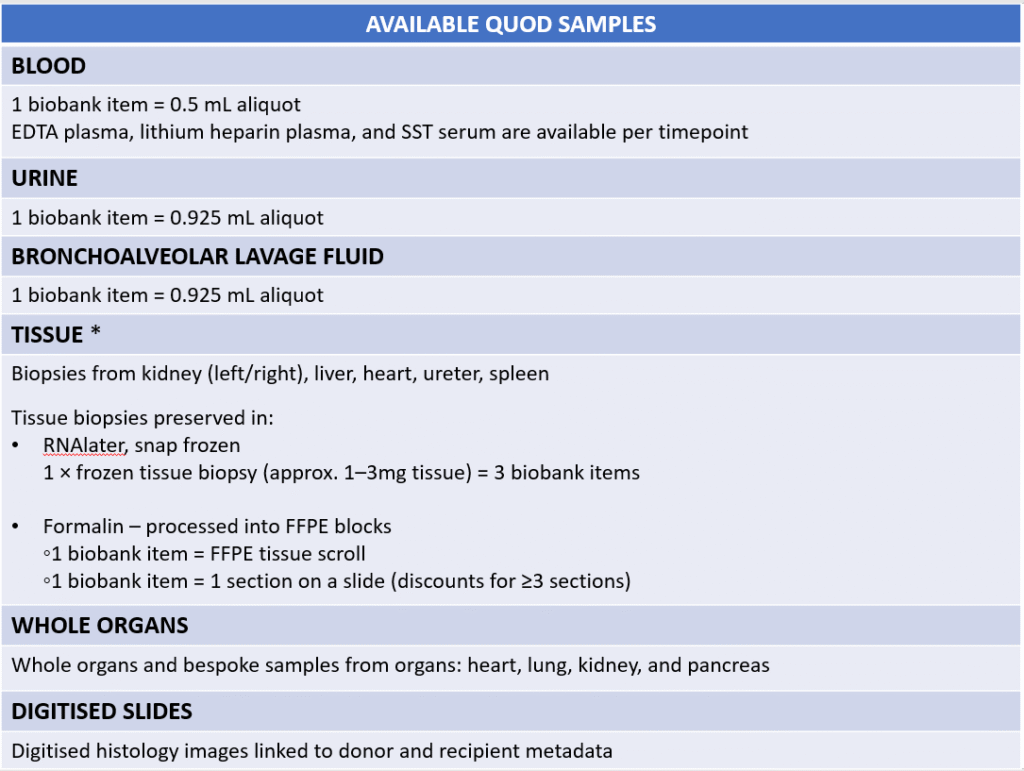
* Available tissue samples are as follows:
Kidney: 3 mm punch biopsy (biopsy types vary since start of sample collection; available samples include: 2 mm punch, 5 mm punch, and core needle biopsies, all halved longitudinally)
Liver: Core needle biopsy (18 G needle, approx. 28 mm length, halved)
Heart: 4 mm punch biopsy from both right and left ventricles (previously 5 mm punch, halved longitudinally)
Ureter: 1 cm length (halved)
Spleen: 1 cm3 (max.) piece
Costs
The QUOD Programme is required to recover some of the cost associated with the running of the biobank. QUOD aims to ensure that material is provided to research projects at the lowest possible cost to researchers.
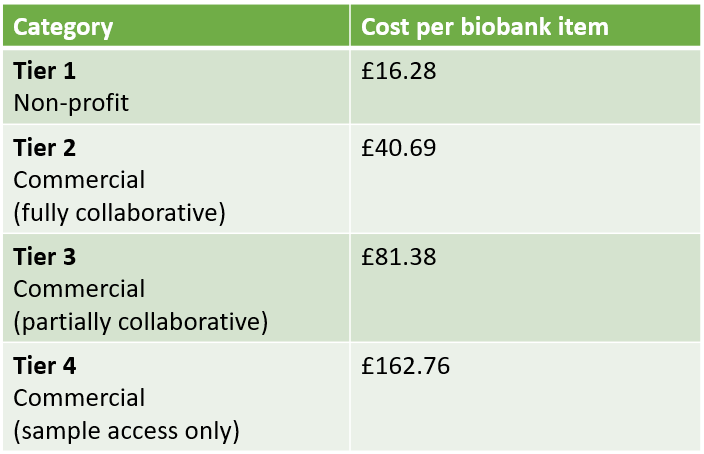
Full details on charges and the cost recovery model are available in the Access Requirements document below.
Access Requirements
Guidance for Researchers
How to Apply
In order to access QUOD samples please complete the Part A Form: Preliminary application, then submit this form via email to quod-research@nds.ox.ac.uk. Part A Form requirements include: a brief outline of the study proposed and the number and types of samples to be requested.
The information will be reviewed by the QUOD team, who will then invite you to provide more detailed information in a Part B Form: Full application for access to the QUOD bioresource. Part B Form applications are then submitted to the QUOD Steering Committee for review.
Once your application has been approved, and the relevant agreements signed, samples matching your selection criteria, together with relevant clinical data (if applicable) will be prepared and dispatched.
Please contact us if you have any questions relating to our sample applications.
Full details of the application process can be found below.
