Q: Which organ donors are included in the QUOD Bioresource?
A: QUOD samples are collected from deceased organ donors after brain death (DBD) and donors after circulatory death (DCD), including paediatric donors over 5 years old.
Q: What biological samples are being collected?
A: Blood, urine, and tissue samples taken at a variety of different time points during the donor management period and retrieval operation. The types of samples included in the QUOD collection may be revisited in response to research needs and in line with QUOD ethics approval.
Q: When are samples collected?
A: The samples are taken from organ donors at four different time points during the donor management period, up until the point of organ retrieval.
Blood samples are collected into EDTA plasma tubes, heparin plasma tubes, and SST serum tubes.
Tissue samples are collected, when appropriate, from left and right kidneys, liver, ureter, spleen, and heart.
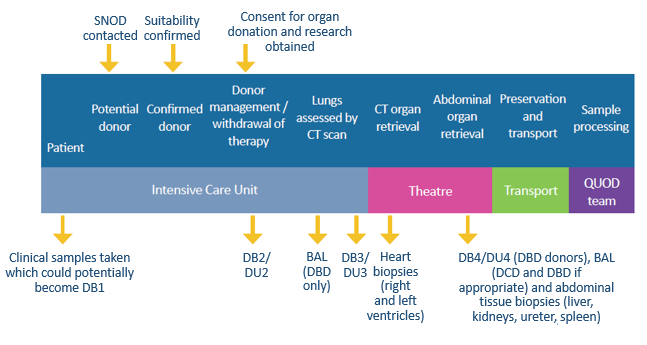
DB: Donor blood, DU: Donor Urine, BAL: Bronchoalveolar lavage
Q: How are the samples processed and stored?
A: Blood, urine, and bronchoalveolar (BAL) samples are centrifuged and supernatants are stored as 0.5ml or 0.925ml aliquots at -80°C. The tissue biopsies are halved at the time of collection and stored in formalin and RNAlater.
Upon receipt at the QUOD biobank, the formalin samples are processed into formalin-fixed paraffin-embedded blocks, allowing a wide range of histological techniques to be exploited. The RNAlater samples are snap frozen in liquid nitrogen and stored at -196°C in vapour-phase liquid nitrogen. This allows highly sensitive techniques to be applied, such as –omics technologies (proteomics, metabolomics, and transcriptomics).
For further details of sample collection, processing, and storage, see the QUOD Access Requirements document.
Q: Why doesn’t QUOD use snap-frozen samples?
A: With QUOD obtaining samples from the majority of all deceased donors in the UK where consent was given for donation and research, the distribution and management of liquid nitrogen across all of our donor hospitals in the UK would be complex, costly, and unduly risky! Early on in the QUOD project, we conducted rigorous audit studies to ensure that RNAlater storage still allows for high-quality omics work. Academic and commercial investigators have been very pleased with the quality and usability of the samples that we have provided.
Q: What are the consequences of taking biopsies?
A: Biopsies are routinely taken in clinical settings. QUOD adheres to strict protocols regarding the biopsy procedure to ensure any potential complications are prevented. Biopsies are taken on the back table after a period of cold perfusion. To ensure appropriate haemostasis and prevent bleeding after reperfusion at time of transplantation, the retrieval surgeon will place a prolene suture at the biopsy site before packaging the liver for transportation. For kidneys, the implanting surgeon sutures the biopsy site prior to transplantation into the recipient.
Q: How many samples are available?
A: The QUOD biobank has collected over 150,000 biosamples from over 8,000 organ donors. Click here to see the latest QUOD statistics report.
Q: Who can access QUOD samples?
A: Researchers from any institution or biomedical company around the world can apply for QUOD samples. The proposals are judged on scientific merit. Click here to find out how to apply.
Q: Why does QUOD charge for samples? How are charges determined?
A: QUOD is a not-for-profit programme. Core funding for the biobank infrastructure is provided by NHS Blood and Transport (NHSBT). Since 2017, the NHSBT Executive Board have required QUOD to recover a proportion of its operating budget. To reach this goal, QUOD has implemented a modest fee for samples.
QUOD aims to keep the cost for a sample for the individual researcher as low as possible. Each year, we review the number of samples distributed in the previous year to make conservative forecasts for the upcoming year, and then divide our cost recovery target figure by the projected number of samples distributed. This generates a ‘cost per biobank item’ that reflects an appropriate percentage of the cost to collect, store, and distribute that biobank item. The QUOD Steering Committee reviews and approves a pricing proposal for the upcoming year.
Additionally, QUOD has implemented a three-tier pricing structure. For details please see the QUOD Access Requirements.
Industrial applicants should contact us at quod-research@nds.ox.ac.uk to discuss options.
Q: How long will it take to receive my samples from application date?
A: In general, we are able to review and approve Part A applications within 2 weeks, and Part B applications within 4 weeks following the submission to the QUOD Steering Committee. In between those applications, researchers will often be in dialogue with the QUOD team to refine sample and data requests. This process can be quite swift if researchers are clear on what they need, but sometimes extends over a month or more if researchers need more assistance in refining their project plans. Once applications are approved, an MTA must be signed before samples can be released. From that point, the provision of samples depends on the overall workload in the QUOD lab and the size of the sample request, but can be as brief as 2 weeks in the right conditions. In general, researchers should plan for at least a 3-month release window from the point of submitting the Part A, with larger or more complex projects taking longer.
Please contact us at quod-research@nds.ox.ac.uk with any further queries.
